Minglei Guo, PhD
Dr. Minglei Guo obtained his Ph.D. in molecular biology from Fudan University, China in 2006. Then, as a post-doctoral fellow in the laboratory of Drs. Yvonne Wan in University of Kansas Medical Center and 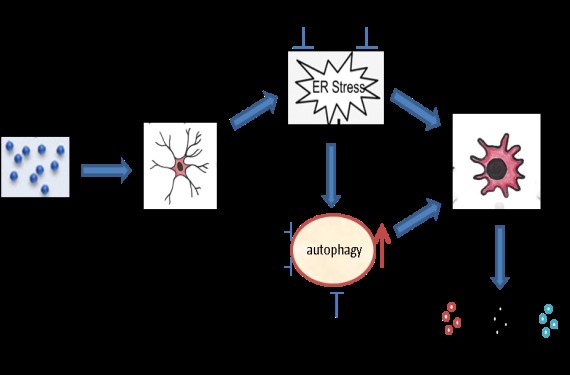 later John Wang in the University of Missouri-Kansas City (UMKC), Dr. Guo focused on understanding the mechanisms involved in cocaine addiction. One of Dr. Guo’s key research achievements involved identifying a novel role for Ca2+/calmodulin-dependent protein kinase and dissecting the mechanisms critical for its binding and phosphorylation of the muscarinic receptor 4 which can be linked with cocaine addiction. In addition to this, his key findings on protein-protein interaction have been critical in our understanding of cocaine-mediated neuronal excitability and behavioral changes. This work was published in the prestigious EMBO Journal in 2010 and importantly, marked his transition into a Research Assistant Professor. Since drug abuse and HIV go hand in hand, in 2013 Dr. Guo joined Dr. Buch’s lab at UNMC and extended his research efforts on exploring HIV-associated neurological complications. Dr. Guo’s recent findings on the role of autophagy, a process critical for maintenance of cellular homeostasis, in mediating cocaine-mediated neuroinflammation, has recently been published in the prestigious Autophagy Journal (2015;11: 995-1009). These findings highlighted that cocaine-mediated induction of autophagy involved upstream activation of two ER stress pathways (EIF2AK3 & ERN1-dependent), which culminated into activation of microglia. Targeting autophagic proteins could thus be considered as a therapeutic strategy for the treatment of cocaine-related neuroinflammatory diseases. Dr. Guo’s current studies are aimed at exploring the role of micro RNAs (miR-124) in drug/HIV-induced addiction/inflammation.
later John Wang in the University of Missouri-Kansas City (UMKC), Dr. Guo focused on understanding the mechanisms involved in cocaine addiction. One of Dr. Guo’s key research achievements involved identifying a novel role for Ca2+/calmodulin-dependent protein kinase and dissecting the mechanisms critical for its binding and phosphorylation of the muscarinic receptor 4 which can be linked with cocaine addiction. In addition to this, his key findings on protein-protein interaction have been critical in our understanding of cocaine-mediated neuronal excitability and behavioral changes. This work was published in the prestigious EMBO Journal in 2010 and importantly, marked his transition into a Research Assistant Professor. Since drug abuse and HIV go hand in hand, in 2013 Dr. Guo joined Dr. Buch’s lab at UNMC and extended his research efforts on exploring HIV-associated neurological complications. Dr. Guo’s recent findings on the role of autophagy, a process critical for maintenance of cellular homeostasis, in mediating cocaine-mediated neuroinflammation, has recently been published in the prestigious Autophagy Journal (2015;11: 995-1009). These findings highlighted that cocaine-mediated induction of autophagy involved upstream activation of two ER stress pathways (EIF2AK3 & ERN1-dependent), which culminated into activation of microglia. Targeting autophagic proteins could thus be considered as a therapeutic strategy for the treatment of cocaine-related neuroinflammatory diseases. Dr. Guo’s current studies are aimed at exploring the role of micro RNAs (miR-124) in drug/HIV-induced addiction/inflammation.
MiR-9 Promotes Microglial Activation by Targeting MCPIP1
 In this study, the authors (Yao H, Yang L, Hu GK, Liao K, Kook YH, Niu F, Liao K, Buch S) demonstrated induction of microglial microRNA-9, a highly conserved microRNA abundantly expressed in the CNS of the developing embryo, following exposure to inflammatory stimuli such as LPS and HIV protein Tat. Our findings implicate miR-9 as a regulator of microglial activation via its suppression of the target anti-inflammatory gene MCPIP1. MCPIP1, a novel anti-inflammatory molecule, was a target of miR-9-mediated microglial activation. Using both pharmacological and genetic approaches, we demonstrated that the
In this study, the authors (Yao H, Yang L, Hu GK, Liao K, Kook YH, Niu F, Liao K, Buch S) demonstrated induction of microglial microRNA-9, a highly conserved microRNA abundantly expressed in the CNS of the developing embryo, following exposure to inflammatory stimuli such as LPS and HIV protein Tat. Our findings implicate miR-9 as a regulator of microglial activation via its suppression of the target anti-inflammatory gene MCPIP1. MCPIP1, a novel anti-inflammatory molecule, was a target of miR-9-mediated microglial activation. Using both pharmacological and genetic approaches, we demonstrated that the 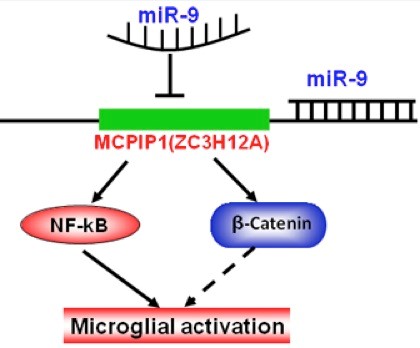 role of NF-κB activation in miR-9-mediated microglia activation. These findings were also validated in vivo using the genetic approach of injecting mice with lentivirus (anti miR-9) into the hippocampus/substantia nigra of mice followed by LPS injection. This resulted in restoration of microglial activation thus validating the role of miR-9 in microglial activation. MiR-9/MCPIP1 axis mediates a regulatory pathway critical for eliciting innate immune responses in microglia. Specific blocking of the miR-9 could thus be considered as a potential therapeutic target for treating neuroinflammatory conditions involving microgliosis.
role of NF-κB activation in miR-9-mediated microglia activation. These findings were also validated in vivo using the genetic approach of injecting mice with lentivirus (anti miR-9) into the hippocampus/substantia nigra of mice followed by LPS injection. This resulted in restoration of microglial activation thus validating the role of miR-9 in microglial activation. MiR-9/MCPIP1 axis mediates a regulatory pathway critical for eliciting innate immune responses in microglia. Specific blocking of the miR-9 could thus be considered as a potential therapeutic target for treating neuroinflammatory conditions involving microgliosis.
Nature Communications (2014) 5:4386
New Faculty
Dr. Siddappa (Sid) Byrareddy was recruited as an Associate Professor in the Nebraska Center for Substance Abuse Research in the Department of Pharmacology & Experimental Neuroscience at UNMC in August 2015. Dr. Byrareddy completed his doctoral training at the premier Jawaharlal Nehru Center for Advanced Scientific Research and the National Institute of Mental Health and Neurosciences in Bangalore India. He completed his postdoctoral training at the Dana-Farber Cancer Institute in Harvard Medical School and was promoted to Instructor within two years. Dr. Byrareddy began his independent  career track as an Assistant Professor at the Emory University School of Medicine. Dr. Byrareddy has a strong background in molecular retrovirology, lentiviral evolution, active and passive immunization and mucosal transmission using primate models. He has over 15 years of research experience and over 50 peer-reviewed publications in the field. Dr. Byrareddy has made significant contributions to his research interests. As a graduate student, he for the first time, showed that tat protein from HIV-1 subtype C is a defective chemokine and provided a model of pathogenesis to explain low levels of dementia in Indians infected with HIV. Furthermore, he has been instrumental in constructing and directing the complete characterization of a number of clade C SHIVs, and has conducted a series of active and passive immunization (involving HIV broadly neutralizing monoclonal/polyclonal antibodies) studies involving mucosal transmission. More recently, his research has focused on understanding gut homing molecules and the development of strategies to protect the gastrointestinal mucosa during HIV/SIV infection. Using anti-α4β7 antibody Dr. Byrareddy’s group has shown that this antibody protects against SIV mucosal transmission. This work was recently published in Nature Medicine. He is currently evaluating interventions aimed at eliminating viral reservoirs from other tissues including the CNS. Dr. Byrareddy is a member of the International AIDS Society, NIH early career investigators group and the global young academy. Dr. Byrareddy has mentored several graduate students, trainees and postdoctoral fellows and has served as PI and co-Investigator on several NIH-funded grants. His ongoing work on opiates and HIV infection makes him ideally suited in understanding the emergence of novel viral variants in Clade C dominant areas of the world. We are thrilled to have Dr. Byrareddy be a part of the NCSAR!
career track as an Assistant Professor at the Emory University School of Medicine. Dr. Byrareddy has a strong background in molecular retrovirology, lentiviral evolution, active and passive immunization and mucosal transmission using primate models. He has over 15 years of research experience and over 50 peer-reviewed publications in the field. Dr. Byrareddy has made significant contributions to his research interests. As a graduate student, he for the first time, showed that tat protein from HIV-1 subtype C is a defective chemokine and provided a model of pathogenesis to explain low levels of dementia in Indians infected with HIV. Furthermore, he has been instrumental in constructing and directing the complete characterization of a number of clade C SHIVs, and has conducted a series of active and passive immunization (involving HIV broadly neutralizing monoclonal/polyclonal antibodies) studies involving mucosal transmission. More recently, his research has focused on understanding gut homing molecules and the development of strategies to protect the gastrointestinal mucosa during HIV/SIV infection. Using anti-α4β7 antibody Dr. Byrareddy’s group has shown that this antibody protects against SIV mucosal transmission. This work was recently published in Nature Medicine. He is currently evaluating interventions aimed at eliminating viral reservoirs from other tissues including the CNS. Dr. Byrareddy is a member of the International AIDS Society, NIH early career investigators group and the global young academy. Dr. Byrareddy has mentored several graduate students, trainees and postdoctoral fellows and has served as PI and co-Investigator on several NIH-funded grants. His ongoing work on opiates and HIV infection makes him ideally suited in understanding the emergence of novel viral variants in Clade C dominant areas of the world. We are thrilled to have Dr. Byrareddy be a part of the NCSAR!
Mentored Fellowship
Ernest Chivero Awarded NIDA-IAS-ANRS Research Fellowship in HIV and Drug Abuse
Ernest, a native of Zimbabwe and Fulbright Scholar recently graduated with a PhD from the University of Iowa where he was studying the delay in HIV disease progression in people infected with both GBV-C (a benign virus) and HIV. He was recently awarded the prestigious postdoctoral research fellowship in HIV & Drug abuse jointly by National Institute on Drug Abuse (NIH-NIDA), International AIDS Society (IAS) & French Research Agency (ANRS) under the mentorship of Dr. Shilpa Buch to perform research in the area of HIV & drug abuse synergy in the context of CNS disease. Despite the successes of antiretroviral therapy in improving the health of millions of people living with HIV-1, paradoxically as these individuals continue to enjoy increased longevity, the prevalence of HIV-associated neurological disorders (HAND) also continues to rise. Ernest’s research is focused on understanding the mechanisms(s) by which residual HIV protein Tat and psychostimulants such as cocaine, co-operative to potentiate inflammatory pathways in the brain microglia, thereby exacerbating symptoms of HAND. His research will contribute to our understanding of the novel molecular pathway(s) critical for neuro-inflammation observed in HIV-infected cocaine addicts, thus providing leads into the development of novel adjunctive therapies for the treatment of HAND and cocaine abuse

