*Note: the UNMC IRB cannot answer questions related to external IRB processes or applications. Contact the external IRB's helpdesk or designated representative directly for assistance.
For questions regarding UNMC cIRB processes or applications, please contact sirb@unmc.edu.
Advarra
- Advarra IRB Application System, CIRBI: https://www.cirbi.net
- Approved UNMC - Advarra Consent Local Context Information Document (11/2/2023)
- Advarra Checklist (optional)
For single-site protocol applications: in the field below in the screen shot (with the x’s).
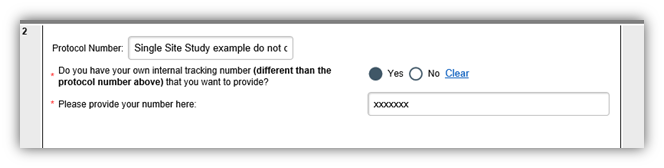
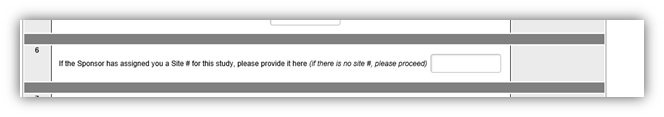
For site only (SSU) applications: add the internal number to the following area on the investigator application:
Contact Information Sheet
- The UNMC IRB recognizes that not all central IRB consent templates have the option to add local study contacts. The cIRB application in RSS now has the option to build a “Contact Information Sheet” to be provided to subjects along with the approved consent documents.
*Note: Older studies within RSS may not have this functionality. The Contact Information Sheet Template may be downloaded for manual completion.
Miscellaneous central IRBs
- Contact the external IRB for information regarding their IRB review process and any applicable IRB application management systems.
- UNMC cIRB Local Context Information Document (11-2-2023)
NCI CIRB
- NCI CIRB Application System, IRBManager: https://nci.my.irbmanager.com/
- Approved Univ of Nebraska Consent Local Context Information Document (09072022)
NMDP
SMART IRB
- SMART IRB https://smartirb.org/ is not an IRB, but a tool used to document reliance between IRBs.
- The IRB representatives from each institution will manage this negotiation. Unless otherwise indicated, nothing is needed from the investigator or study teams to fulfill this requirement.
WCG
- WCG IRB Application System, WCG IRB Connexus: https://identity-connexus.wcgirb.com
- Approved U of Nebraska - WCG Consent Local Context Information Document (1/12/2024)
*Note: If requesting revisions to the template consent language, the U of Nebraska – WCG ICF Checklist must be completed and signed by the study’s lead UNMC IRB representative.


