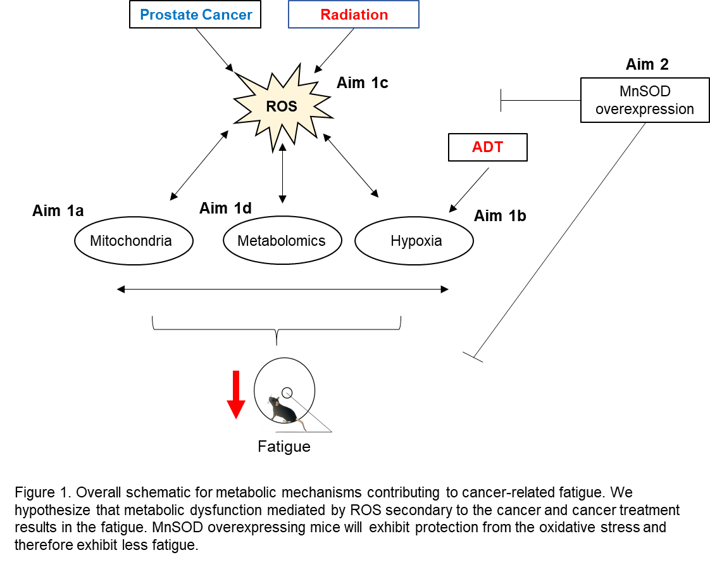
| Purpose: There are no specific treatments for cancer-related fatigue (CRF), attributed to a lack of knowledge of the biologic mechanisms contributing to this pathology. The proposed preclinical study is needed to enhance access to tissues unavailable in the clinical setting so we can advance understanding of the biology underlying CRF. Therefore, we will test our central hypothesis that metabolic mechanisms drive the pathobiology of CRF. Approach: C57Bl/6 WT or TgMnSOD mice will be implanted with RM-1 prostate tumor cells. Mice will be randomized to receive androgen deprivation therapy consisting of daily enzalutamide (25 mg/kg) intraperitoneal injections for 7 days + pelvic radiation for 3 days for a total dose of 24 Gy. After completion of radiation, mice will be placed in individual cages equipped with voluntary running wheels for assessment of fatigue. Target tissues (blood, muscle, brain, and prostate) will be used to investigate metabolic mechanisms (mitochondrial quality and function, hypoxia, metabolomics, and oxidative stress). All biologic data will be correlated to our primary outcome of fatigue. Expected Outcomes: This study provides the unique opportunity to take the bedside (our previous clinical studies) back to the bench (animal model of CRF) to further understand the mechanistic underpinnings of CRF. We predict that we will observe differences in metabolic profiles between the fatigued and non-fatigued mice. We also predict that the MnSOD overexpressing mice will exhibit increased voluntary wheel running activity, demonstrating that protecting the mitochondria from superoxide damage results in less fatigue. |
 Funding: This research is supported by a UNMC internal pilot award from the Fred and Pamela Buffett Cancer Center and IDeA CTR. |

