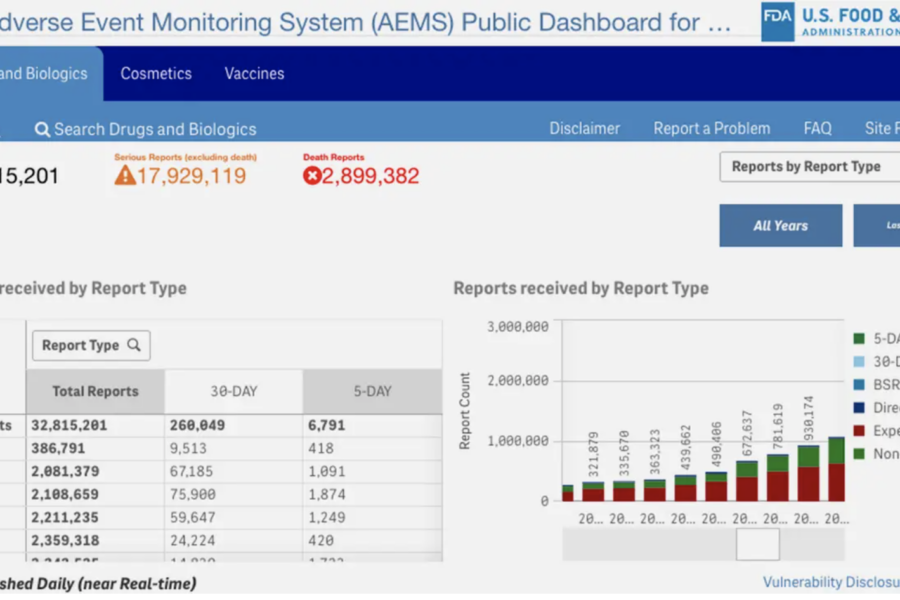
MedPage Today System will replace safety databases monitoring drugs, devices, food, and more. “The FDA’s previous adverse event reporting systems were outdated and fragmented and made important data difficult to access,” FDA Commissioner Marty Makary, MD, MPH, said in the release. “These clunky systems also wasted millions of taxpayer dollars and created blind spots in our postmarket surveillance of products ranging from drugs and vaccines to cosmetics … Starting today, the FDA will have a single, intuitive adverse event platform that will better serve agency scientists, researchers, and the public.”
The agency emphasized the improvements in data aggregation and opportunities for research that the new system will provide. “Adverse event reports submitted to the FDA for drugs, biologics, vaccines, cosmetics, and animal food can be displayed in a single streamlined dashboard,” the release said. “By the end of May 2026, AEMS will contain real-time adverse event reports for all FDA-regulated products, consistent with meeting agency obligations not to release individually identifiable patient or consumer information.”