Samuel Cohen, MD, PhD
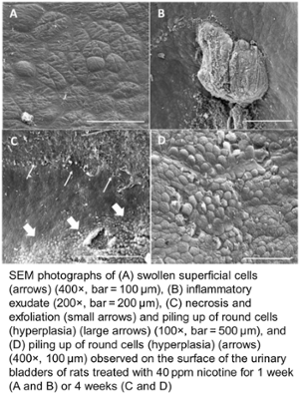
My research involves several aspects of carcinogenesis, with an emphasis on urinary bladder as a model system in rodents and extrapolation between rodent models and human diseases. We have postulated that agents increase cancer risk by either directly interacting with DNA or increasing cell proliferation in appropriate target cells, allowing for more opportunity for spontaneous mutations to occur during DNA replication. Genotoxic chemicals, such as aromatic amines, nitrofurans, and nitrosamines require metabolic activation, DNA adduct formation, and mutagenesis. Numerous nongenotoxic chemicals have been identified, enhancing carcinogenesis by increasing urothelial cell proliferation, including arsenic, sodium salts, amino acids, calculus-forming chemicals, and phenolic chemicals. Increased proliferation occurs either by direct mitogenesis, such as by high doses of propoxur, or more commonly by cellular toxicity and consequent regenerative hyperplasia, such as occurs with formation of calculi by chemicals like uracil, by processes involving more subtle formation of urinary solids, such as following high doses of sodium saccharin, or by chemical cytotoxicity and regeneration such as by inorganic arsenic, nicotine, diuron or pulegone. Based on our investigations, it is unlikely that chemicals acting by formation of urinary solids pose a carcinogenic hazard to humans. Cytotoxic chemicals might be relevant to humans but involve a threshold, high level of exposure. Our studies involve various aspects of toxicology, basic chemistry, cell kinetics, electron microscopy, pathology, in vivo and in vitro bioassays, renal physiology and molecular biology. In addition to the urinary bladder, we have applied similar approaches to investigations on liver, lung, kidney, intestine, testes, and vascular tumors.

