KNOCKOUT MOUSE
MGE core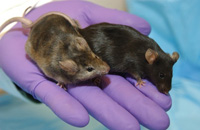
- Generation of Chimeras
- Breed Chimeras to produce F1 pups: obtain tail clips
- Transfer of mutant F1 mice as well as all Chimeras to PI laboratory
- Advice on breeding strategies and strain maintenance
PI laboratory
- DNA preparation, genotyping and identification of mutant pups
- Analysis of gene knockout mice
ES cells and blastocysts - the fine print…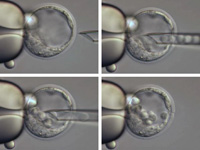
- MGE will only use confirmed recombinant ES cell clones for blastocyst injection. As an ES cell clone is propagated for blastocyst injection, the morphology of the cells will be documented, and presence (or absence) of cells with differentiating (instead of stem-) morphology will be logged. If excessive differentiating morphology (i.e. more that 25% of ES cell colonies in the culture show a differentiated phenotype) is encountered, the ES cell clone in question will not be used for blastocyst injection.
- Fresh media will be used for each injection experiment, and the batch numbers of media as well as all additives will be logged. Batch number of hormones used in superovulation or stimulation will be documented, together with number of mice, time of injection, and amount of hormone used for each procedure.
- MGE will produce at least 30 viable pups from blastocyst injection experiments for a given gene targeting project. To achieve this goal, MGE will perform at least two blastocyst injection sessions for each project. Depending on the number of blastocysts that were obtained, successfully injected, and transferred to foster mothers in the first experiment, the second injection may use a different recombinant ES cell clone than the one used in the first experiment.
- MGE expects to obtain at least 10 chimeras from 30 offspring. Typically, 10 chimeras are sufficient to achieve germline transmission of the targeted allele.
- MGE will classify blastocyst embryos collected for injection: (i) injectable blastocyst; (ii) injectable morula; (iii) hatched blastocyst; and (iv) unusable/dead. Numbers for these categories will be logged. These data will be correlated with the hormone batch numbers on a monthly basis for quality control purposes.
- For each recombinant ES cell clone, MGE will inject blastocyst embryos – category I and II only - with the recombinant cells, with an average of 8 ES cells per blastocyst embryo. MGE will strive to generate at least 120 blastocysts that carry ES cells and are alive after an expansion period of 30-60 minutes as determined by microscopic inspection. Only such live blastocyst embryos will be transferred to foster mothers. Assuming an implantation rate of 30%, this will lead to the production of ~ 30-40 offspring, a sufficient number to generate chimeras, i.e. animals that are a mixture of host-derived as well as ES cell-derived cells and tissues. Should a lower implantation rate be experienced, more blastocyst injections with the respective ES cell clone will be carried out.
- MGE will use the 'number of viable pups produced' as the primary benchmark, not the number of embryos that were injected, or the number of injection experiments that were conducted.
- If no chimeric animal can be identified in 30 offspring, the ES cell clone in question will no longer be used for blastocyst injections, and another ES cell clone will be used. MGE starts with parental ES cell lines that have proven to produce germline-transmitting chimeras in an effort to ensure successful chimera production and germline transmission. In case of such a failure, MGE will report its efforts to the Chair of the MGE Advisory Committee; the Committee will determine whether failure to produce chimeras among 30 offspring derived from ES-cell injected blastocysts would be due to the individual ES cell clone in question, due to the nature of the targeting construct, or due to MGE procedures.
- Initial breeding of chimeras to test for germline transmission of the targeted allele (investigator will genotype) will be performed by MGE to insure that (i) chimeras are fertile, and (ii) chimeras are sufficiently experienced in their contact with female mice so that chances for accidents (i.e. wounding or killing of the chimeric male by the female mouse) are minimized. For a gene targeting experiment, three to five chimeras will be set up to breed until they successfully sire two litters each, after which chimeras (together with any agouti F1 offspring) are transferred to the Investigator's laboratory.

